Supply Chain Management integrated Services
At NEWEDGE, we offer a range of supply chain management integrated services in the pharmaceutical sector. With dedicated teams to cater to all your diverse needs, we ensure that we become your one-point solution provider for a host of products and services, which can also be tailored as per the unique requirements and priorities of our clientele.
CONTRACT DEVELOPMENT
We are a trusted name in the industry for contract development of APIs and products in FDF forms for robust supply chain management. With a team of experts that manage your project, we ensure that every available resource is leveraged to provide you with the best of the solutions for contract development. The factors that enable NEWEDGE Overseas to optimise this process include robust interdisciplinary coordination, benefiting from an extensive network of partnerships to track the best possible options and having unlimited access to technologies and processes.
A dedicated team for the analysis and evaluation of market potential is another factor that helps us cater to country specific requirements. With committed teams that ensure that all the projects are managed in a timely and efficient manner, we have created a niche for ourselves in the contract/product development sector in the global pharmaceutical industry.


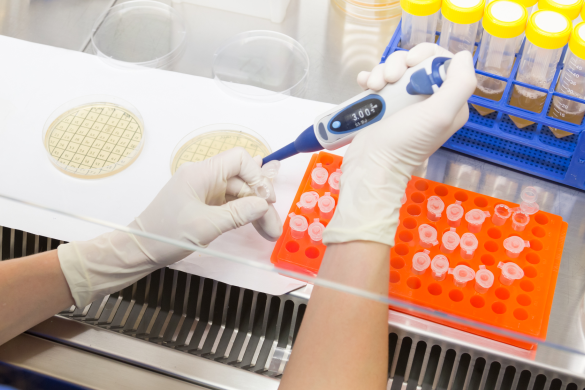
CONTRACT MANUFACTURING
By leveraging our extensive network of partners, we offer solutions that are standardised as well as are tailored as per your specific needs and requirements to become your trusted collaborators in the field of contract manufacturing. Our services encompass manufacturing of APIs, FDFs and nutraceuticals in different dosage forms and intermediates. With a dedicated team that boasts of commendable expertise as well as enviable experience, we offer need-based solutions as per client requirements with efficacy and speed.
Our complementary service packages also include product sourcing, quality management, ensuring regulatory compliances, rigorous quality checks, and other supply chain services, fulfilled by in-house NEWEDGE experts. We provide access
to technologies that are unique, distinct, and differentiated, expertise in chemical handling, and varied production capabilities and scalability, in association with trusted CRO and CDMO partners. With our capability to handle multi-ton synthesis, we can help you reduce the turnaround time and the quality of your supplier portfolio. With all these, we become your one-point solution for all your requirements, reducing your outsourcing qualification interventions for superior supply chain management.
- We provide API and advanced intermediate synthesis exclusively complying with the latest regulatory requirements as per the highest quality standards and cGMP.
- We also manufacture early-stage intermediates and building blocks with our non-GMP capabilities and expertise.
- From start-ups and MSMEs to globally established corporations, we cater to all.
- Leveraging the latest technologies, we are innovative in our research and chemical experimentation.
- From grams to tons, our production scale exceeds volumes of your expectations.
- Our scientifically driven project management team evaluates, manages, and implements different projects as per the client’s expectations.
- With respect to FDFs, we provide every kind of contract manufacturing services irrespective of the dosage form.
QUALITY MANAGEMENT
With a series of regulatory compliances in place, at NEWEDGE Overseas we take over the responsibility of quality management for you. With processes in place for guideline and regulation tracking, we fulfil this responsibility and ensure that for you, the process becomes seamless and cooperative. The superior quality of the products we deliver also extend their shelf-life and their effectiveness. This helps us deliver superior quality products within a stipulated time frame. The customer hence benefits not only on quality parameters, but is assured that no compromises have been made when it comes to the cost or speed of delivery. With this, our promise of quality extends itself to accommodate the promise of timely delivery and cost effectiveness.

SUPPLY CHAIN MANAGEMENT
With a robust mechanism in place, NEWEDGE Overseas undertakes due-diligence of the supply chain and manages all due processes with commendable ease of operations. We have a strong network of over 400 partners. This evidently results in competitive prices for our clients and into timely delivery of consignments, and thus, into premium quality services for our clients. This, more often than not, helps our customers expedite their production/sales, accelerating the rate of supply, elevating quality (by increasing shelf life) and cost effectiveness. Moreover, our supply chain resilience extends to hard-to-find APIs also, as with tech empowerment and a robust network, we are able to maintain a speedy supply chain.
SOURCING OF HARD-TO-FIND APIs
With a wide network of partners, we specialize in procurement of rare APIs on customer demand. Our extensive directory of products, both APIs and FDFs includes some rare and hard-to-find APIs. Nevertheless, if our customers are in need of an API that is not a part of our portfolio, we help find those by employing market intelligence and a tech empowered system that leverages various softwares and connections. Moreover, through collaborations on various levels (low, medium and upper), we ensure that the rare APIs sourced withstand all quality checks and come at a competitive price. Our promise of quality and competitive price also stands for these specially sourced products.


ANALYTICAL METHOD DEVELOPMENT
At NEWEDGE Overseas, we offer services pertaining to Analytical Method Development and Analytical Method Validation, both of which are critical processes for drug development. Our services include drug assessment that encompasses testing and providing information regarding the potency, stability, bioavailability and effect of various drugs. We also undertake characterizing Analytes to ascertain their biological and chemical properties. These services facilitate cost-optimisation and help minimize influence on efficiency, precision and accuracy of drugs.
LICENSING OPPORTUNITIES FOR PRODUCT DEVELOPMENT
At NEWEDGE Overseas, we provide both in and out licensing opportunities for product development, which are essential considering the ambitious approach of entities from the Indian pharmaceutical sector in order to accelerate its growth on national and international levels. The services we offer include licensing of intellectual properties, patenting of products and processes, facilitating use of trademark/brand name, copyrighting of processes and technologies, providing technical advice and assistance, opportunities to launch differentiated or specialised products, providing access to new technologies and manufacturing processes and formulating long-term strategies that would help widen global reach.


PACKAGING AND LABELLING
Branding, packaging and labelling of APIs, FDFs, Nutraceuticals, Excipients and Intermediates, in various forms, customized as per the client requirements. Our services include packaging and labelling in a temperature and humidity controlled environment, mass-packaging, designing and branding services, packaging regulatory control and flexible packaging. We are equipped with the necessary tools and an advanced facility through which we manage complete control over the products’ quality, efficacy and longevity.