Successful supply chain management is an essential tool for pharmaceutical companies to boost company sales and lower expenses. To provide quality products at a reasonable cost, companies need to maintain an adequate supply chain across the country and globe. Following are some salient pointers on supply chain management:
What is Supply Chain Management?
Supply chain management is defined as a fusion of the processes starting from procuring raw materials, manufacturing of goods to providing finished products to the customers. It is a systematic business model which integrates supply and demands within stakeholders and customers to gain competitive advantages in business. The primary goal of any supply chain management process is enhancing accessibility, quality, and affordability.

How does the Supply Chain Work in the Pharmaceutical Industry
The Pharmaceutical supply chain includes multiple government agencies, hospitals, clinics, drug manufacturers, drug distributors, pharmacy chains, retailers, research organizations, and the FDA. As a result, expectations from the supply chain managers in the pharmaceutical industry are very high as drugs supplied inappropriately can damage customer loyalty and public health. The flow of the supply chain in the pharmaceutical industry can be described in the following steps:
- The drug product is manufactured at the company site
- It is transferred to the wholesale distributors
- Stocked at pharmacies for retail or online purchase
- Subjected to price negotiations and processed through quality and utilization management screens by pharmacy benefit management companies
- Dispensed by pharmacies and ultimately taken by patients

Challenges of Supply Chain Management
Here are some significant challenges which can hinder the supply chain and should not be overlooked:
- Maintaining the quality of drugs
- Timely delivery of products
- Sustaining the desired temperature throughout the transport for particular drugs
- Inventory and warehouse management
- Order and shipment management
- Taking care of expiry of products
- Managing the delivery of drugs during any shortage
Digitalisation as per the current needs
A robust supply chain is essential to meet the dramatic changes of the pharmaceutical industry, and hence digitalization is the key to transformation. As a result, pharmaceutical companies must evaluate their existing supply chain strategies to improve efficiency and ensure optimum product quality. The cornerstone of a multi-enterprise supply chain is a digital business network. Digitalization electronically links all of the supply chain partners across the cloud. It makes it possible for them to work together when pharmaceutical supply chain partners are related and offers end-to-end visibility and an incentive for multiple business interactions to collaborate.
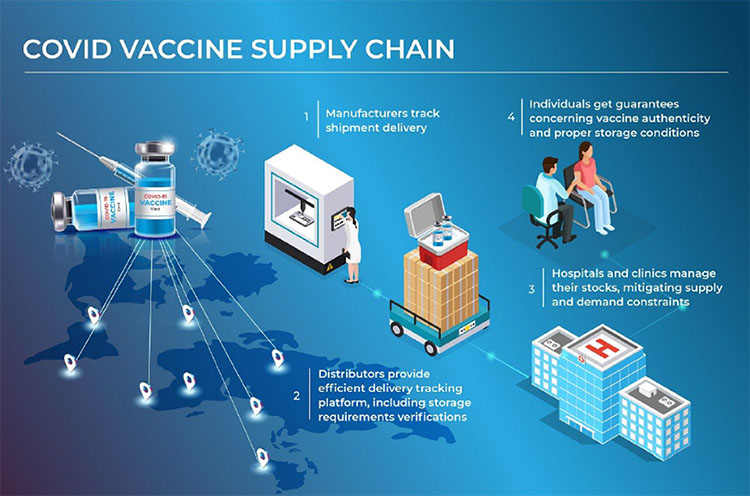
How did Covid 19 Pandemic change the dynamics of the Supply Chain?
The covid 19 pandemic has exposed some supply chain deficiencies that must be addressed and corrected for the future. For example, during the lockdown in several countries, the supply of raw materials and APIs were highly affected, resulting in delayed production of finished dosages. Therefore, the industry must not rely on only a few sources of supply to avoid shortages and crises. Today, pharmaceutical companies are vigilant and paying close attention to their suppliers, and maintaining constant contact through digital technologies as a learning lesson.
Conclusion:
The performance of pharmaceutical companies can vary based on the efficiency of supply chain management. Companies have to identify the risks related to the supply chain and prevent them from avoiding business risks. Hence, it is essential to maintain adequate supply chain management in the Pharmaceutical Industry. With a robust mechanism in place, NEWEDGE Overseas undertakes due diligence of the supply chain and manages all due processes with commendable ease of operations. Moreover, our supply chain resilience extends to hard-to-find APIs. Moreover, with tech empowerment and a robust network, we can maintain a speedy supply chain. Kindly contact NEWEDGE Overseas for premium quality services.